53
Research Trials
20
Peer-reviewed publications
16
Clinical Conditions
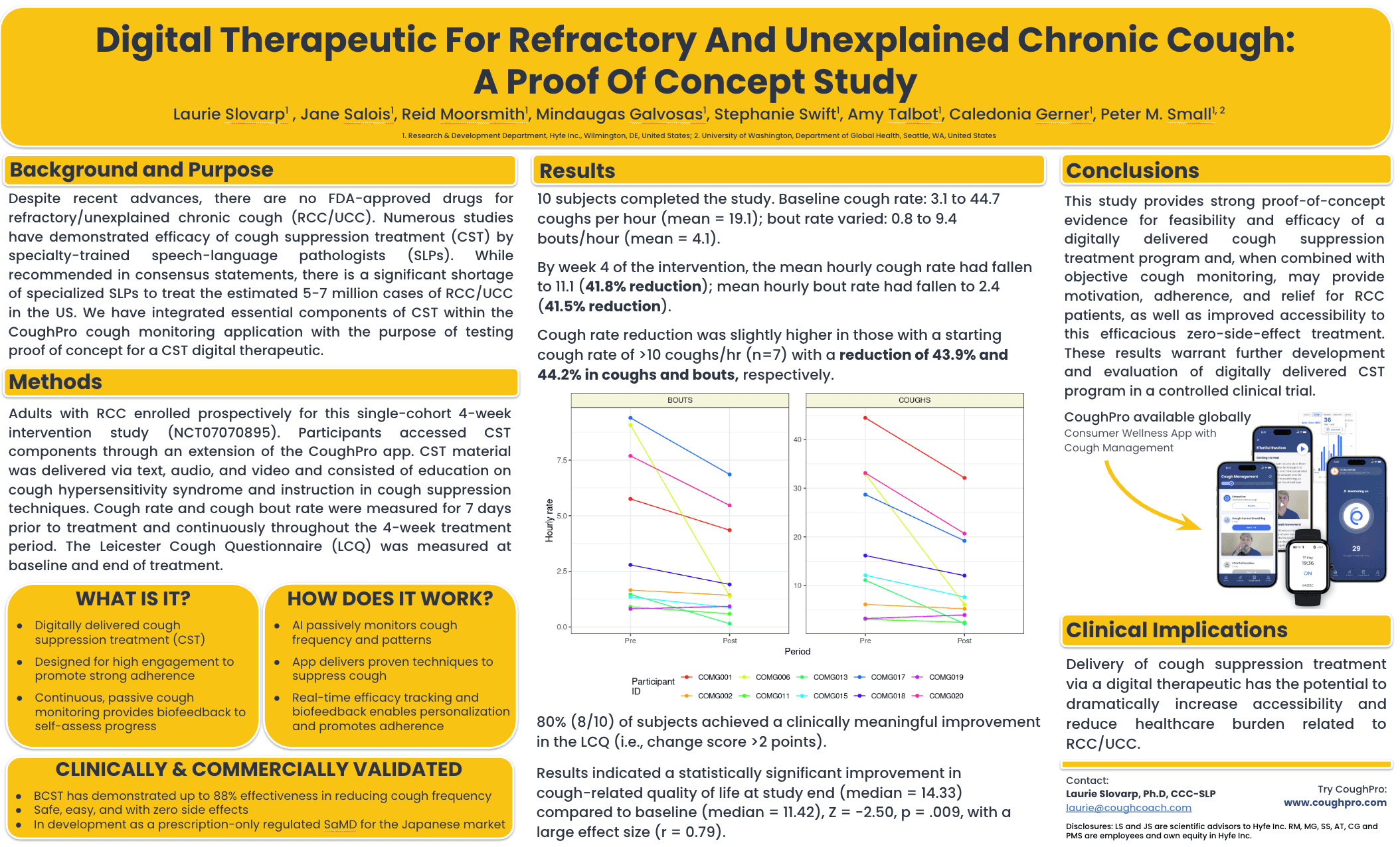
At CHEST 2025, held in Chicago, Illinois, Laurie Slovarp, PhD, CCC-SLP, professor at the University of Montana and certified speech pathologist, presented a poster on the development of a digital therapeutic designed to improve access to behavioral cough suppression therapy for patients with refractory chronic cough.

A large-scale trial examining the effect of azithromycin on the relationship between oesophageal function and cough as evaluated by Hyfe's cough monitoring technology in respiratory disease is feasible and acceptable to patients.

This study used Hyfe's wearable cough monitor during a 7-day run in, 28-day treatment, and 14-day follow-up period in patients with chronic bronchitis.

Periods of intense coughing (termed bouts, epochs or bursts) are particularly problematic for some coughers and may not be reflected by simply counting the number of coughs per day. This study explored how varying the definition of bouts yield different impressions of cough severity.
01.10.2024

Background:
The hydration status of surface liquid in the larynx and trachea may regulate peripheral cough receptors.
Hypothesis:
Topical hypertonic divalent salts (HDS) buffered to alkalinity provides relief for refractory chronic cough.
We conducted an exploratory, single-blinded controlled study in 12 chronic cough patients to examine cough-suppression efficacy of a nasally delivered HDS composition (SC001) at pH 8 or pH 9, compared to a normal saline (NS) control. Subjects were monitored continuously using the Hyfe CoughMonitor smartwatch for one week of baseline, one week of NS, and one week of SC001.
Baseline cough rates ranged from 4 to 41 coughs/hr with a mean visual analog score (VAS) of 65±17. Adjusting for control, SC001 demonstrated efficacy at suppressing cough by 15% from Day 1 (p=0.015) and 23% from Day 3 (p=0.002) overall (n=12). Efficacy was highest in subjects treated with SC001 pH 9 (n=5), with a 25% cough-suppression observed from Day 1 (p=0.03) and 35% from Day 3 (p=0.02). Lower efficacy was observed for SC001 pH 8 (n=7), with 9% (p=0.08) and 16% (p=0.02) suppression observed from Day 1 and Day 3 respectively. Hourly cough counts correlated with VAS scores (ρ = 0.266, p-value = 0.015) at baseline and increased with treatment (ρ = 0.507, p-value = 0.000002). No adverse events were reported.
Alkaline (pH 9) HDS aerosol is a promising treatment for refractory chronic cough.