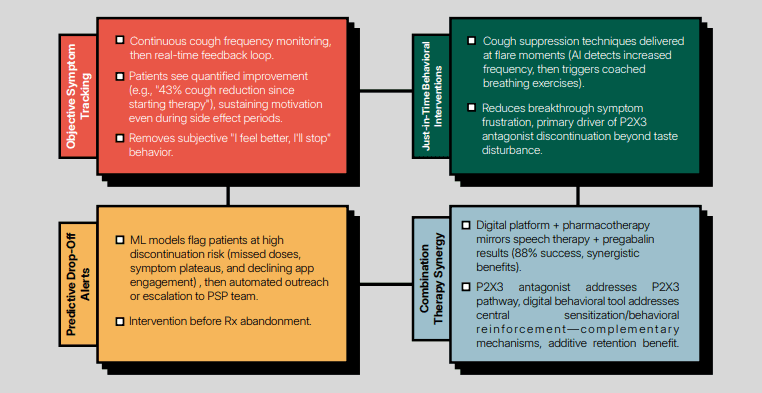
Objective cough monitoring for drug development and clinical trials
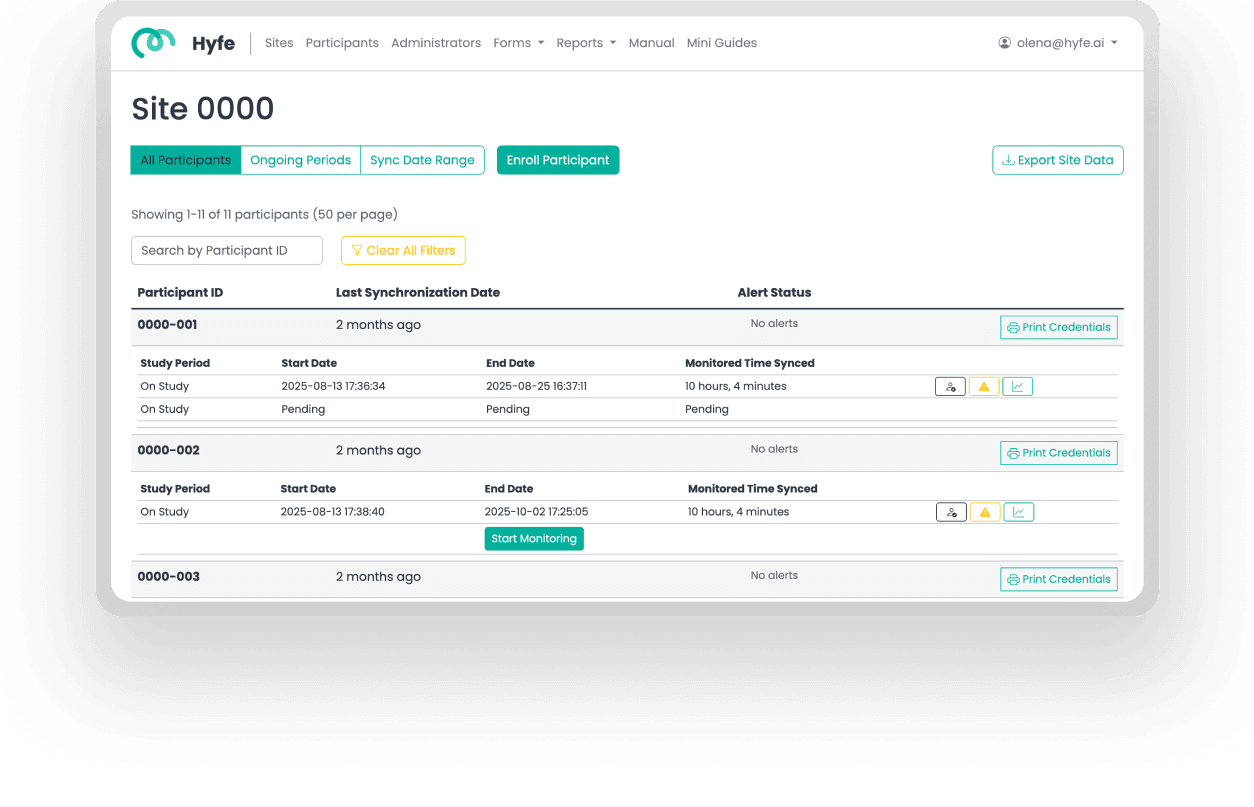
Hyfe’s AI-powered cough monitoring is unobtrusive for patients and can monitor continuously for days, weeks or months. For the first time ever, the secrets of longitudinal cough patterns can be unlocked.